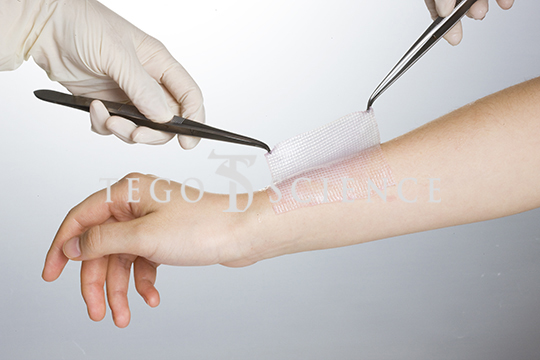
Kaloderm®
[Prescription Drug] [National Health Insurance Reimbursement]
- Description
Kaloderm® is the world's third allogeneic cell therapy product for healing wounds.
It is cultured from the master and working cell banks that contain the keratinocytes from the foreskin of a circumcised infant. We have the technology to propagate the cells almost indefinitely, meaning that Kaloderm® will always be made from the single source and maintain the same quality.
Kaloderm® is mass-produced on a regular basis and can be stored frozen at -60℃ for 36 months. Since its MFDS approval in 2005, over 400,000 sheets have been applied and is reimbursed by the Korean National Health Insurance.
DFU indication was added in 2010 and approved for National Health Insurance reimbursement in 2019.
- Indications & Clinical use
1. Promote re-epithelialization of deep 2nd degree burn
2. Promote the healing of non-infected
diabetic foot ulcer wounds
- Active Ingredient
Allogeneic Keratinocytes
- Size
56cm², 9cm²
*A more detailed description can be downloaded to reveal the product documentation.
Download